IN8bio ($INAB) : Undervalued Oncology Play or Capital Crunch Disaster?
Is this a value trap headed for dilution and oblivion—or a deeply mispriced recovery play for investors who can live with binary risk?
If you look at the chart of IN8bio ($INAB), you see a catastrophe. Following a 1-for-30 reverse split in June 2025 and a relentless grind lower, the market has effectively written this company off. With a market cap hovering around $8 million—trading below its Q3 cash balance of $10.7 million—Wall Street is assigning a negative enterprise value to its clinical pipeline.
But if you look at the data, you see something closer to a miracle.
In the graveyard of oncology—Glioblastoma (GBM) and high-risk AML—IN8bio is posting survival curves that don’t look like historical experience. In AML, 100% of treated patients remain in complete remission with 100% progression-free and overall survival at one year (median follow-up ~20.1 months). In GBM, a repeat-dose cohort achieved more than double the progression-free survival seen with the standard Stupp protocol. These are small, early-phase, single-arm trials, but the magnitude of the signals is hard to ignore.
This is the classic “Biotech Dilemma”: a company with science that could change the world, trapped in a capital structure that is suffocating it. This Deep Dive explores whether $INAB is a value trap—or the ultimate recovery play for the patient investor.
Science Deep Dive: Why This Is Not Just “Another CAR-T”
To understand the value here, you have to understand why IN8bio is fundamentally different from the CAR-T therapies (like Yescarta or Kymriah) that have flooded the market.
The Problem with Standard CAR-T (Alpha-Beta T Cells)
- Blindness: Conventional CAR-T uses alpha-beta (αβ) T cells. These cells are “blind” unless they’re genetically forced to see a specific target (like CD19). If the tumor hides that target (antigen escape), the therapy fails.
- Rejection risk: You cannot take αβ T cells from a donor and put them in a patient without complex gene editing, or they will attack the patient’s healthy tissues (graft-versus-host disease, GvHD).
- The “cold” tumor problem: In solid tumors like glioblastoma, the environment is hostile. To get T cells to work, you usually have to wipe out the patient’s immune system first (lymphodepletion), dose the cells, and hope they survive. But you can’t keep giving full-dose chemotherapy at the same time, or you kill the expensive CAR-T cells.
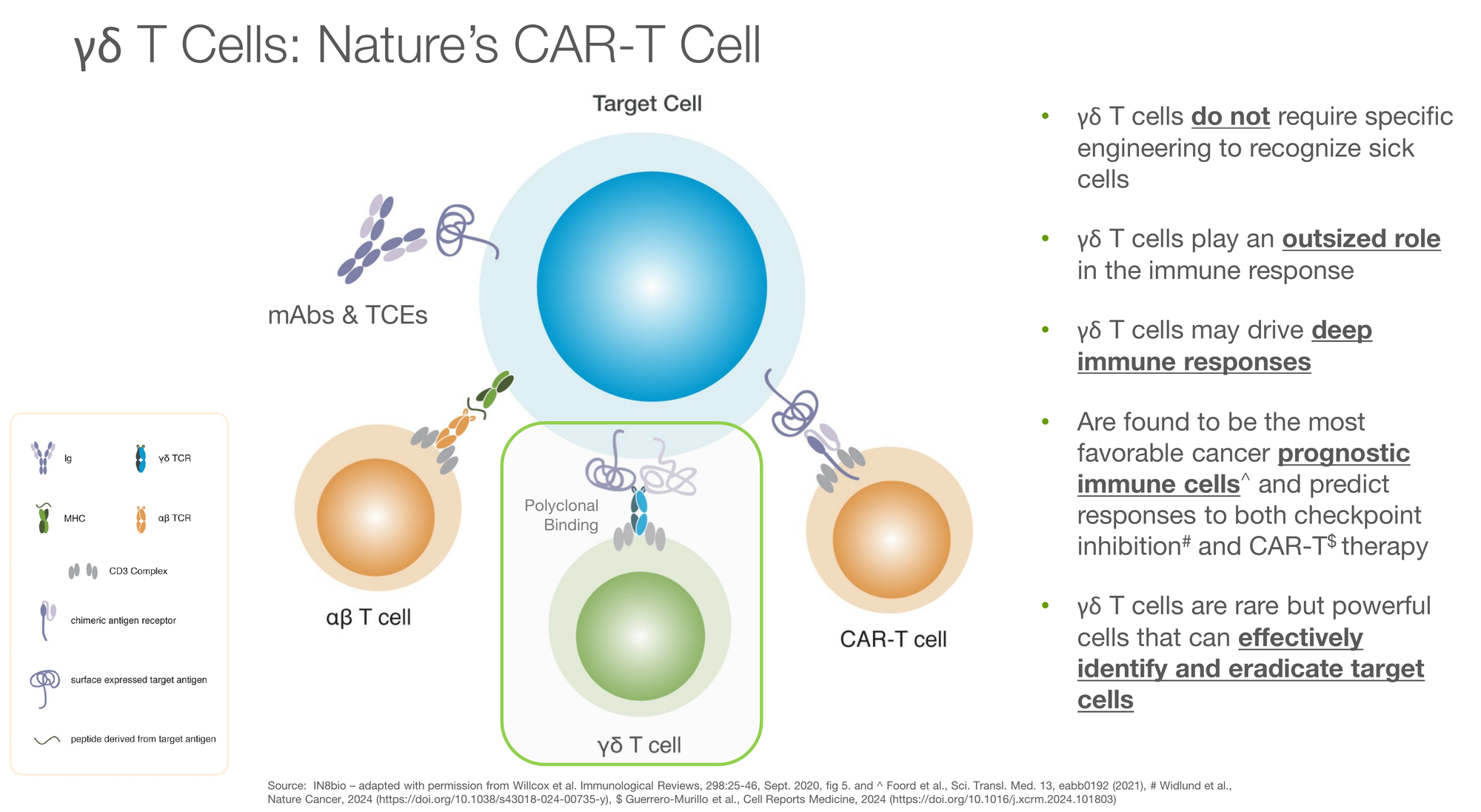
The IN8bio Solution: Gamma-Delta T Cells + DRI
IN8bio uses gamma-delta (γδ) T cells. These are the “special forces” of the immune system. They rely less on a single engineered target and naturally hunt down patterns of “stress signals” on cancer cells (such as NKG2D ligands).
The “killer feature” is Drug Resistant Immunotherapy (DRI). IN8bio genetically modifies γδ T cells with a gene called MGMT-P140K.
- What it does: It makes the immune cells resistant to chemotherapy (specifically temozolomide).
The Synergy Loop (The “One-Two Punch”)
- Chemo first: You hit the patient with chemotherapy. This kills bulk tumor cells and causes surviving cancer cells to “panic,” displaying stress ligands on their surface.
- Cell infusion: You infuse IN8bio’s MGMT-engineered γδ T cells into that same environment.
- Chemo-resistant hunters: Because they’re chemo-resistant, these cells survive the toxic milieu. They see the “panic signals” on the tumor and attack.
- Room to expand: The chemotherapy clears out parts of the patient’s old immune system, creating space for IN8bio’s cells to expand and patrol.
Bottom line: Standard CAR-T often forces you to stop chemo to save the T cells. IN8bio’s approach tries to use chemo itself to supercharge the T cells.
The Glioblastoma Signal (And Why It Was Paused)
Glioblastoma (GBM) is the widow-maker of biotech. For roughly 20 years, the standard of care (Stupp protocol) has barely budged: about 6.9 months of progression-free survival (PFS).
IN8bio’s lead solid-tumor asset, INB-200, tried to break that ceiling.
In their Phase 1 trial, patients receiving multiple doses of their genetically modified γδ T cells achieved a median PFS of 16.1 months:
- Standard of care: 6.9 months
- INB-200 repeat-dose cohort: 16.1 months (+133% improvement vs Stupp)
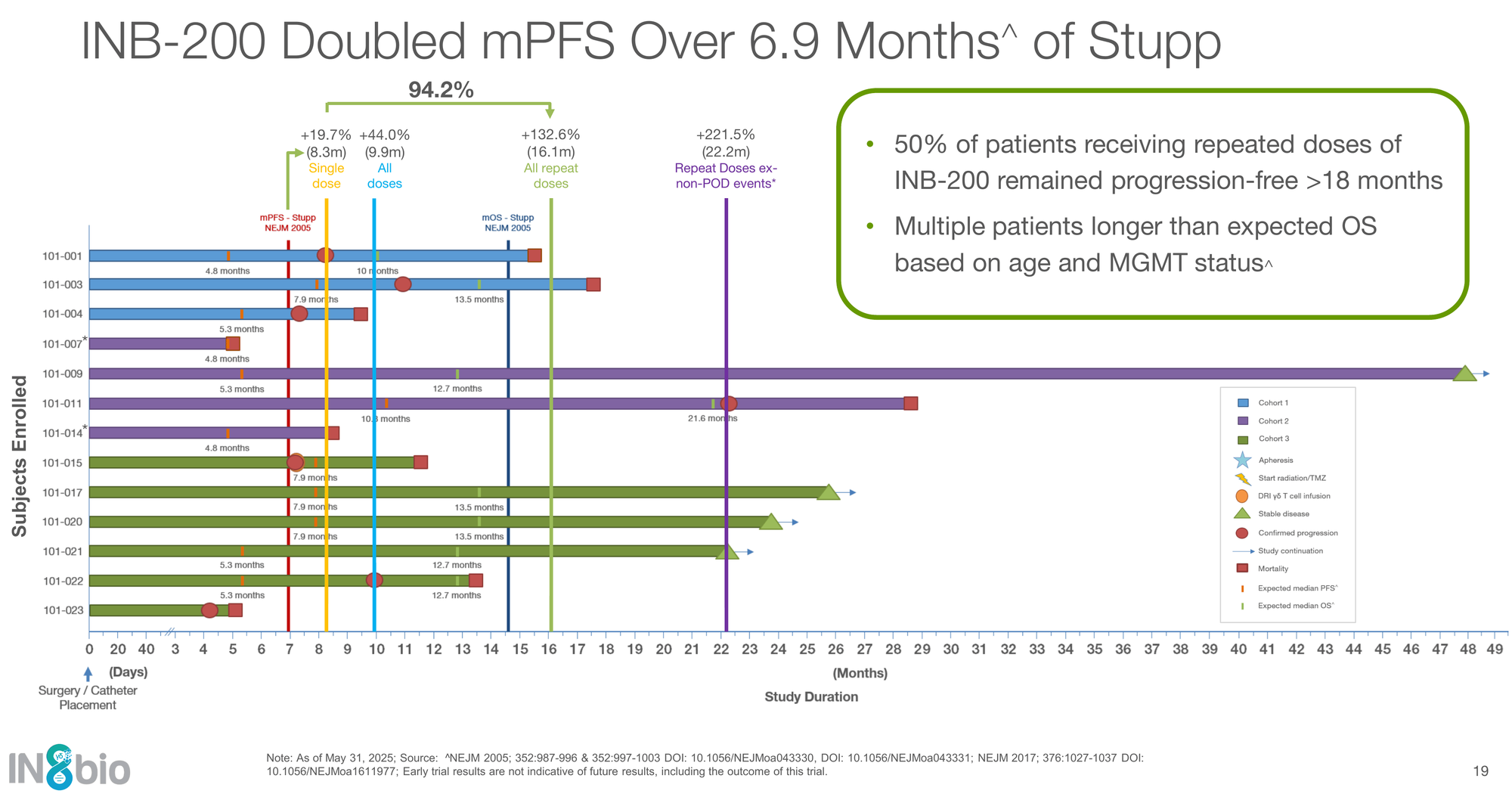
Even more striking is the “long-term survivor” signal. As of mid-2025, multiple repeat-dose patients remained alive and progression-free, including one patient beyond four years—extraordinary durability in a disease where recurrence within a year is typical.
The caveat: This is a small, single-arm Phase 1 study. All comparisons are against historical controls, and GBM is notorious for trial-to-trial variability. The data are hypothesis-generating, not yet definitive.
Why was INB-400 paused?
Despite this signal, IN8bio paused the Phase 2 trial of the successor candidate, INB-400, in September 2024—not because the drug failed, but because they didn’t have the capital to push both GBM and AML programs forward. Management chose to prioritize INB-100 and conserve cash.
The opportunity: INB-400 is effectively “shovel-ready” and sitting on the shelf. A partnership deal to co-develop the GBM program is arguably the single biggest non-dilutive catalyst that could re-rate the stock overnight.
The “Perfect” Curve: INB-100 in Leukemia
With GBM paused to save cash, the company went “all in” on INB-100, their allogeneic (donor-derived) therapy for acute myeloid leukemia (AML) patients undergoing transplant.
The data here look cosmetically perfect so far.
As of the January 2025 data cut presented at the TCT Meetings:
- 100% of treated AML patients remain in complete remission.
- 1-year PFS and OS are both 100%.
- Median follow-up: 20.1 months.
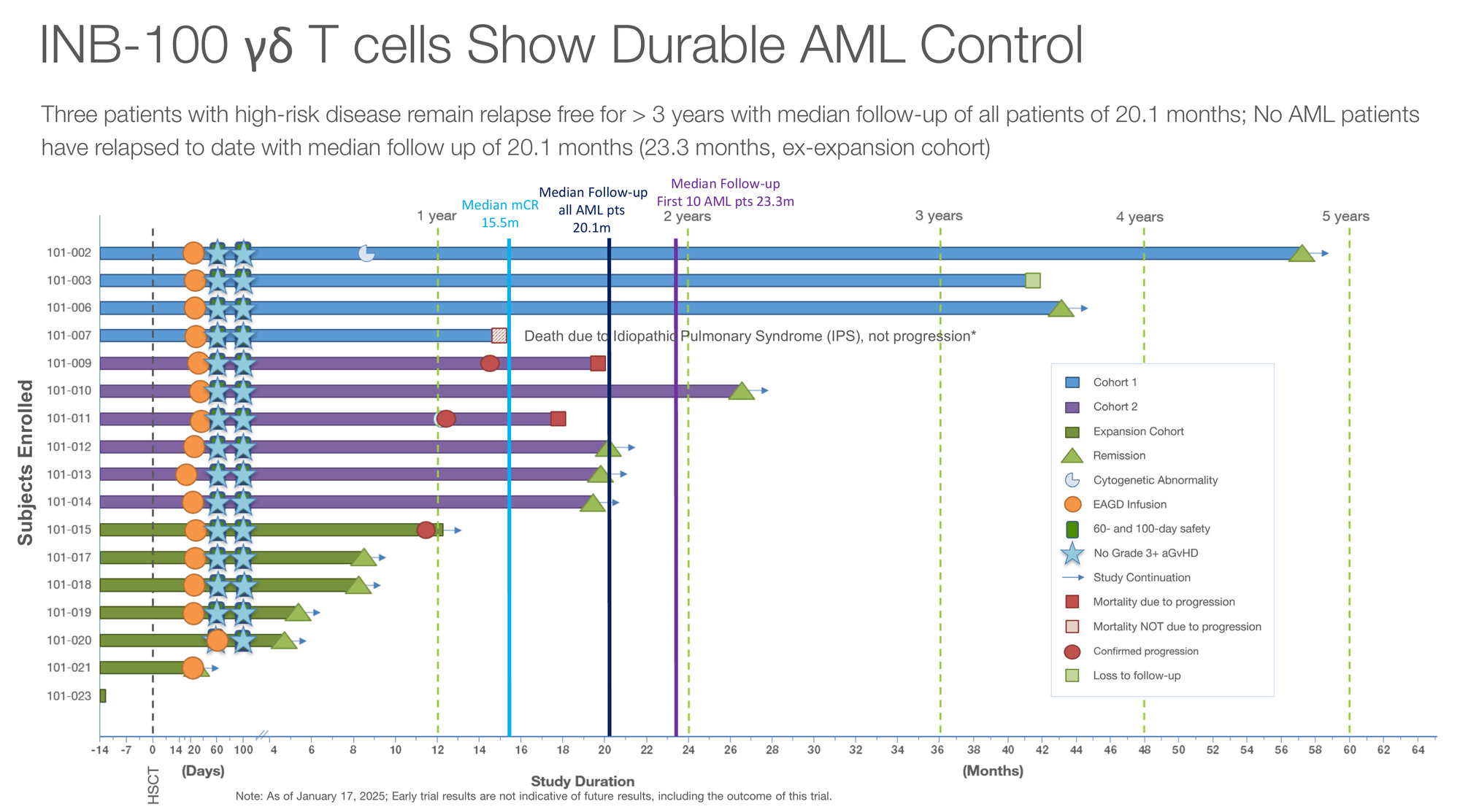
In high-risk AML, post-transplant relapse is the primary cause of death. To have zero relapses observed at nearly two years suggests that IN8bio’s γδ T cells may be acting as a “living security system”, hunting down residual leukemic blasts that chemotherapy and conditioning missed.
The program has now expanded to The Ohio State University, signalling that despite the cash crunch, top-tier academic centres see enough promise in the data to open the trial.
Again, the usual caveats apply: it’s a small, single-arm dataset, and long-term follow-up will tell us whether the curve stays this clean.
The Risks: Why Is It Trading at $1.60?
We have to look at the bear case with open eyes. $INAB is a nanocap for a reason.
- Liquidity crunch: As of Q3 2025, the company had about $10.7M in cash. Based on current burn, the company has cash runway into mid-2026 at best. That’s a critically short leash. They’re burning roughly $10.6M over nine months (≈$3.5–4M per quarter), even after a major workforce reduction and pipeline cuts.
- Warrant overhang: The company has funded itself through multiple equity financings and warrant deals (including Series C warrants). This creates a ceiling on the stock price: when good news hits, warrant holders can exercise and sell into strength, suppressing sustained rallies.
- Execution risk: They need a deal. Without a Big Pharma partnership, substantial licensing cash, or an outright buyout, they cannot afford the registrational trials needed for approval in GBM or AML.
- Unproven science: IN8bio pioneers gamma-delta T-cells, a modality distinct from proven alpha-beta CAR-Ts. While the sector needs innovation, investors are notoriously risk-averse toward unverified mechanisms. Capital currently views this as a "science experiment," applying a steep novelty discount until the approach is further verified.
Upcoming Catalysts: The “Make or Break” Window
The next 6–9 months are critical for IN8bio.
- SNO Annual Meeting (Nov 19–23, 2025): Updated long-term survival data from the GBM program (INB-200/INB-400). If the survival curves hold or improve with longer follow-up, the asset becomes much harder for potential partners to ignore.
- Autoimmune & TCE Readouts (late 2025–2026): INB-619, IN8bio’s CD19-targeted γδ T-cell engager, has already shown deep B-cell depletion in Lupus samples with a cleaner cytokine profile than traditional CD3-based engagers. Additional preclinical data from the γδ TCE platform are expected as they try to position themselves in the “immunology plus oncology” sweet spot.
- Partnership watch (H1 2026): Management has been clear that current cash only takes them into mid-2026 and that they are actively seeking additional financing and collaborations. In practice, that usually means some combination of asset partnering, licensing, or strategic transactions.
Conclusion: A Lottery Ticket with Scientific Validation
IN8bio is not a “safe” investment. It is a distressed asset play.
The bull case:
You are effectively buying a Phase 2-ready GBM asset and a clinically validated AML asset for roughly the value of the cash on the books. If they sign a partnership deal for INB-400—or secure a broader strategic transaction—the stock could re-rate dramatically given the tiny float and negative enterprise value.
The bear case:
They fail to secure funding or a partner, the runway runs out in mid-2026, and shareholders face massive dilution, program shutdowns, or insolvency.
Final verdict:
$INAB is a tier-one scientific innovator trapped in a tier-four financial vehicle. For those with a high risk tolerance, this is a stock to keep on the radar. The data are too good to simply disappear; the real question is whether today’s equity holders will still be around to benefit when someone finally decides to pay for it.
Watch for: a partnership or licensing announcement around the paused GBM program. That’s the match that could light the fuse.
Disclaimer
The information provided on this website is for informational purposes only and should not be construed as
financial, investment, legal, or professional advice. While efforts are made to ensure accuracy, no guarantee
is given regarding completeness or reliability. Visitors should conduct their own research or consult a qualified
advisor before making any decisions. External links are provided for convenience and do not imply endorsement.

