The Biotech Edge: Analyzing 8,000 Catalysts to Find the Alpha
Data-driven biotech investing: Biostockinfo reveals how clinical catalysts, anticipation run-ups and Breakthrough Therapy designations create alpha for independent retail investors.

The mainstream financial narrative—peddled by major networks and institutional reports—frames biotechnology as a sector driven purely by fundamental science and binary outcomes. While not entirely wrong, this view is incomplete. For the retail investor, the "institutional playbook" is often a trap, designed for liquidity rather than maximization of returns.
At Biostockinfo.com, our mandate is different. We don't answer to investment banking arms or compliance departments. We answer to the data.
Our proprietary analysis of over 8,000 clinical catalysts from January 2023 through November 2025 reveals a market that is not "efficient," but rather structurally mispriced. By understanding where these inefficiencies exist—specifically in anticipation run-ups and regulatory designations—independent investors can find a statistical edge that the "smart money" often overlooks.
Part 1: The Anticipation Arbitrage
A common misconception is that price strength leading into a data readout signals insider confidence. Our data suggests a different reality: the market pays a premium for uncertainty in small-caps, while discounting maturity in large-caps.
The Small-Cap Speculative Premium
When we isolate the 30-day pre-catalyst window, a clear inverse correlation appears between clinical stage and anticipation returns.
- The Data: Preclinical and Phase 1 assets consistently demonstrate the highest average run-ups, with aggregate data showing mean returns exceeding 40% in the month prior to a catalyst.
- The Driver: This is not necessarily a reflection of clinical probability, but of market structure. These equities are typically low-float, high-beta assets. The mere possibility of a positive outcome creates a liquidity vacuum that drives prices higher on relatively low volume.
- The Implication: For the astute trader, the "trade" is often the volatility itself, not the scientific outcome.
The Late-Stage Efficiency Trap
Contrast this with Phase 3 or PDUFA events. Our datasets indicate that anticipation returns for these headline events are often flat to negative (averaging near -0.5%).
Why does the market punish late-stage anticipation?
- Institutional Hedging: By Phase 3, institutional ownership is high. Funds are risk-managing, often selling calls or trimming positions to hedge against binary failure.
- Valuation Gravity: The company is no longer valued on a dream, but on a looming commercial reality.
Data Spotlight: The Inefficiency Gap
The graph below visualizes the stark contrast in pre-catalyst behavior. Note how the "speculative frenzy" cools dramatically as assets mature into Phase 3.
Average 30-Day Anticipation Return by Clinical Stage
(Note: This chart tracks the mean run-up from T-30 to T-1 days before the catalyst date)
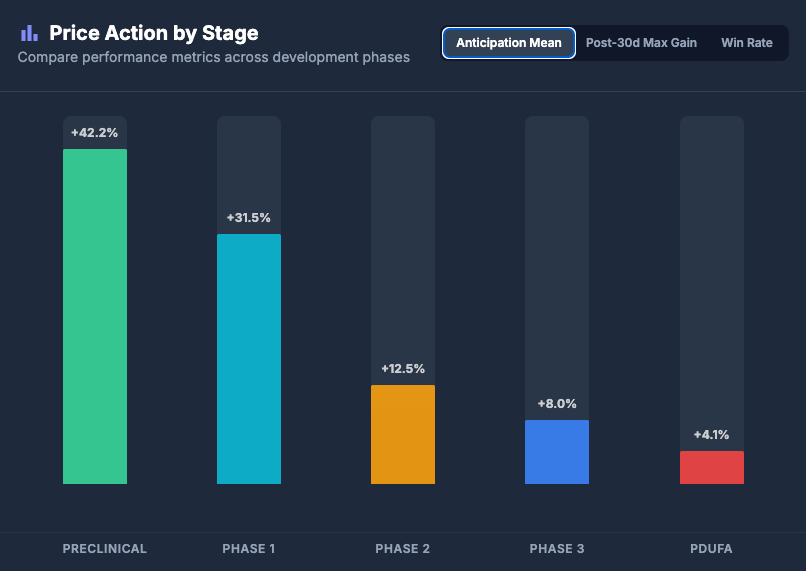
Source: Biostockinfo.com Internal Analysis
(over 8,500 Catalysts from recent data between Jan 2023 - Nov 2025)
Phase 1 includes : Phase 1, 1a, 1b
Phase 2 includes : Phase 2, 2a, 2b, 1/2
Phase 3 includes : Phase 3, 2/3
Analyst Takeaway: Do not chase Phase 3 run-ups expecting momentum. The statistical "alpha" exists in the early-stage speculation phase—provided one exits before the binary event converts potential energy into kinetic risk.
Part 2 : The Binary Paradox
The "Catalyst Day" is the sector's defining feature. However, our analysis of immediate post-catalyst returns suggests the "Law of Diminishing Returns" applies heavily to drug development.
Assessing the Pop
Counter-intuitively, Phase 3 and PDUFA wins often generate lower immediate percentage returns than early-stage surprises.
- Pricing Efficiency: A positive Phase 3 readout is frequently "priced in" to the market cap. A multi-billion dollar entity may only move 10-20% on approval.
- The Phase 2 Sweet Spot: The most explosive revaluations occur at Phase 2. Here, the company transitions from "science experiment" to "viable asset." The valuation gap between these two states is where the multi-bagger returns are statistically clustered.
The Post-Catalyst Drift
For the investor focused on capital accumulation rather than day-trading, the Post-30d window offers a compelling signal.
- The Trend: Stocks that deliver unequivocal positive data often experience a "post-catalyst drift." Once the event-driven volatility settles, fundamental funds—which require weeks to update models and approve buy orders—begin to accumulate.
- Strategy: If the data validates the platform, the dip following the initial spike is often a buying opportunity for a medium-term hold.
The chart below isolates only the positive outcomes (winners) to show how they perform during the month after the news drops.
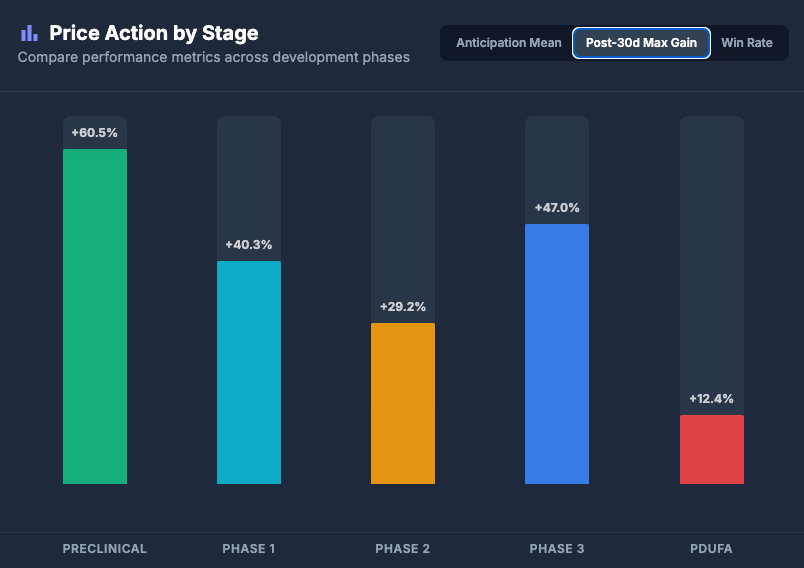
(Note: This chart tracks the mean maximum performance anytime between T+1 to T+30 days for companies with positive outcome)
Analyst Takeaway: Phase 3 winners often deliver strong gains over the subsequent 30 days in this dataset, whereas PDUFA outcomes show the weakest average returns, as they tend to be fully priced in beforehand.
Part 3: The Regulatory Alpha – FDA Breakthrough Therapy Designation
If anticipation plays are about market structure, long-term holds must be about probability. In a sector where 90% of drugs fail, we need a filter to identify the assets the FDA is actively shepherding toward approval.
The FDA Breakthrough Therapy Designation (BTD) is that filter.
Recent industry reports, specifically a comprehensive analysis by Jefferies (as reported by BioSpace), highlight BTD not just as an accolade, but as a material de-risking event.
The Statistical Edge
Breakthrough Therapy Designation vs Standard Drug Development
| Metric | Standard Drug Asset | BTD Asset |
|---|---|---|
| Historical Approval Rate | <10% (from Phase 1) | ~75% |
| FDA Review Timeline | 10 Months | 6 Months |
| Priority Review Status | Discretionary | 94% Rate |
Source: Jefferies / BioSpace Article
- 72% - 75% Approval Rate: Historical data indicates BTD assets have an approval rate nearing 75%. In a sector where the baseline is often single digits from inception, this designation dramatically shifts the probability of success (POS) in the investor's favor.
- The Velocity Factor:
- 94% of BTD drugs are granted Priority Review.
- This statutory mechanism reduces the FDA review clock from the standard 10 months to 6 months.
- Financial Impact: Accelerating time-to-market by four months does not just save cash; it pulls forward peak sales, materially increasing the Net Present Value (NPV) of the asset.
The "Partner" Dynamic
When the FDA grants BTD, the relationship shifts from "Regulator-Applicant" to something closer to a partnership. The designation confers intensive guidance from senior FDA staff, allowing companies to address deficiencies before they become Rejections (CRLs). For the investor, a BTD is a signal that the regulator perceives a high unmet need and the drug has shown substantial clinical promise.
Conclusion: The Independent Approach
The biotech market rewards those who can distinguish between market mechanics (anticipation runs) and fundamental value (BTD assets).
You can consider few strategies from these findings:
- Exploit Volatility: Utilize the high-beta nature of early-stage (Phase 1/2) anticipation runs, but recognize them as liquidity events, not investment theses.
- Avoid the Crowd: Be wary of the "Phase 3 Crowded Trade." The risk-reward is often skewed against the retail buyer.
- Follow the Regulator: Weight portfolios toward Breakthrough Therapy Designations. The 75% historical win rate—backed by Jefferies' data—provides the statistical floor necessary for long-term outperformance.
- Buy the Drift: Accumulate confirmed winners in the quiet period (Post-30d) as institutional money slowly positions.
We provide the data. You execute the trade.
Disclaimer
The information provided on this website is for informational purposes only and should not be construed as
financial, investment, legal, or professional advice. While efforts are made to ensure accuracy, no guarantee
is given regarding completeness or reliability. Visitors should conduct their own research or consult a qualified
advisor before making any decisions. External links are provided for convenience and do not imply endorsement.

